 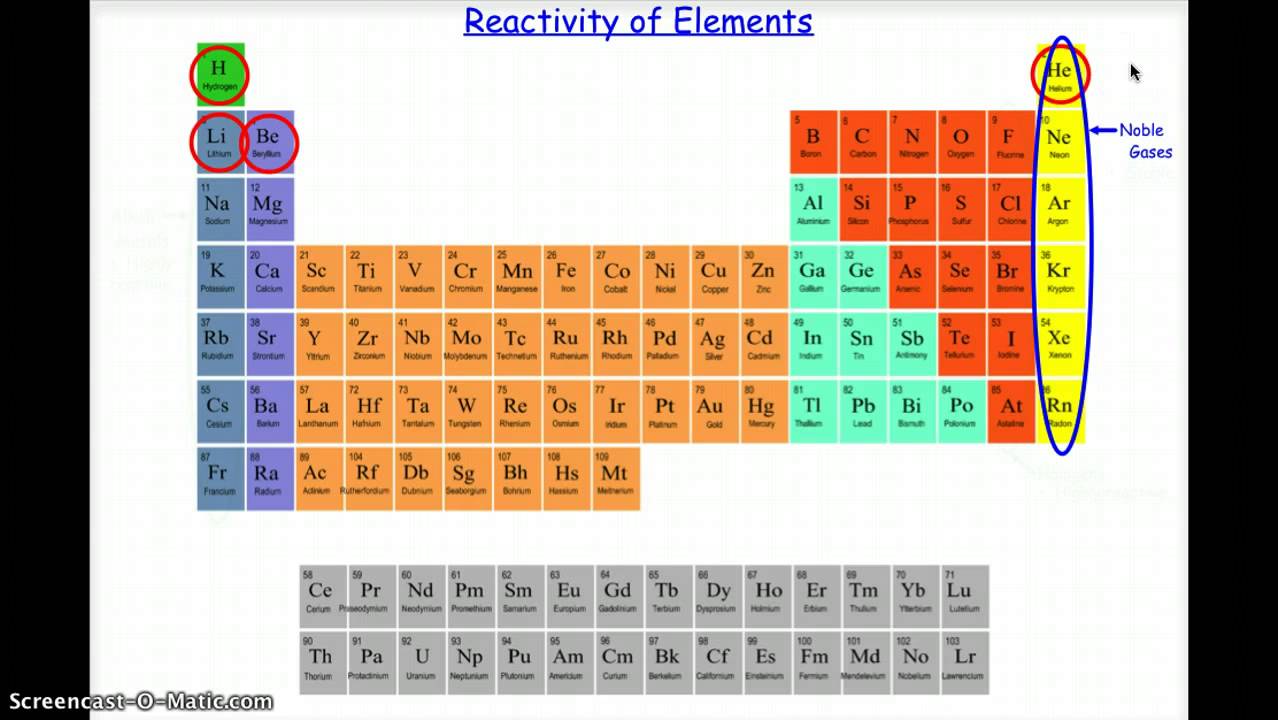
In the modern periodic table, elements are arranged in order of atomic number in periods and groups. Gethin: So, the further down the table you go the more reactive they become. Practical - determining a reactivity series Mendeleev made an early periodic table. Bubbles of hydrogen gas are given off, and a white precipitate (of calcium hydroxide) is formed, together with an alkaline solution (also of calcium hydroxide. Calcium, for example, reacts fairly vigorously with cold water in an exothermic reaction. So, the reason we didn't see the reactions of those last three metals is that they are so reactive the explosion would be far too big. Strontium and barium have reactivities similar to lithium in Group 1 of the Periodic Table. Four major factors affect reactivity of metals: nuclear charge, atomic radius, shielding effect and sublevel arrangement (of electrons). Suitable for gifted and talented students aged 14-18 years in the UK. Binod Shrestha (University of Lorraine) The elements within the same group of the periodic table tend to exhibit similar physical and chemical properties. Miss Armit: No, it's the second rarest element on Earth. A resource for chemistry teachers to explore the patterns of reactivity in groups 1 and 7 of the periodic table. The students will arrange the diagrams and their. This activity is great to use once your students understand that the identity of an atom can be determined by its number of protons, and that in a neutral atom the protons equal the electrons. That's not an element that you hear a lot of. This activity involves simple graphics of the atoms of elements 1-20 of the Periodic Table. We saw there lithium, then sodium and then potassium, but you didn't see rubidium, caesium and francium. As you can see the metal's get increasingly more reactive as you go down this group. Gethin: It's fair to say that you love the Group 1 metals, don't you. Well, sodium is below lithium in group one and the reactivity of the alkali metals increases as you go down the group, so potassium is even more reactive. So let's try it with another Group 1 metal - sodium.Īs you can see it reacts far more vigorously then lithium. And can you see the little purple trail there? That universal indicator is showing us that the water now contains an alkali in this case - lithium hydroxide. It's transferring its energy to its surroundings by heating. Well, you can see it's reacting with the water straight away - it's steadily fizzing away there. 
And if I put some universal indicator in, like this, you'll see that it's green and that means it's neutral.īut let's see what happens when we put the lithium in. And do you see how quickly it tarnishes, how quickly it's changing colour? Here we've got some water. Here I've got some lithium and I'm just going to cut a little bit off. When a Group 1 element reacts with water it produces a metal hydroxides solution and hydrogen gas. They're soft and shiny when freshly cut, but they quickly tarnish as the metal reacts with oxygen in the air. Let's take a look at some Group 1 metals then - also known as alkaline metals.įran: Group 1 elements are called the alkali metals. Magnesium is the eighth most abundant element in the Earth’s crust, and calcium is the fifth.Gethin: And today we're going to be looking at the periodic table. Calcium is found in chalk, limestone, gypsum and anhydrite. The main minerals in which magnesium is found are carnellite, magnesite and dolomite. Instead, they are widely distributed in rock structures. These elements are all found in the Earth’s crust, but not in the elemental form as they are so reactive. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
